


A potential manufacturing error has prompted health officials to issue a recall on a common blood pressure medication, warning that it could cause some concerning side effects.
Recalls are common across a number of different industries, as it feels like there's one every week in the car world and you're bound to have been affected by a recall with an item of food or drink that you've bought in the past.
These are obviously concerning – although not always as dangerous as they sound – as they are often prompted by worrying discoveries that could lead to health issues or injury, but it's understandably even more concerning if a recall occurs with a form of medication.
Advert
As reported by the Independent, that's exactly what's happened with one of the most common forms of blood pressure medication, as health officials have issued a recall for capsules produced within a single batch due to a potential manufacturing error.
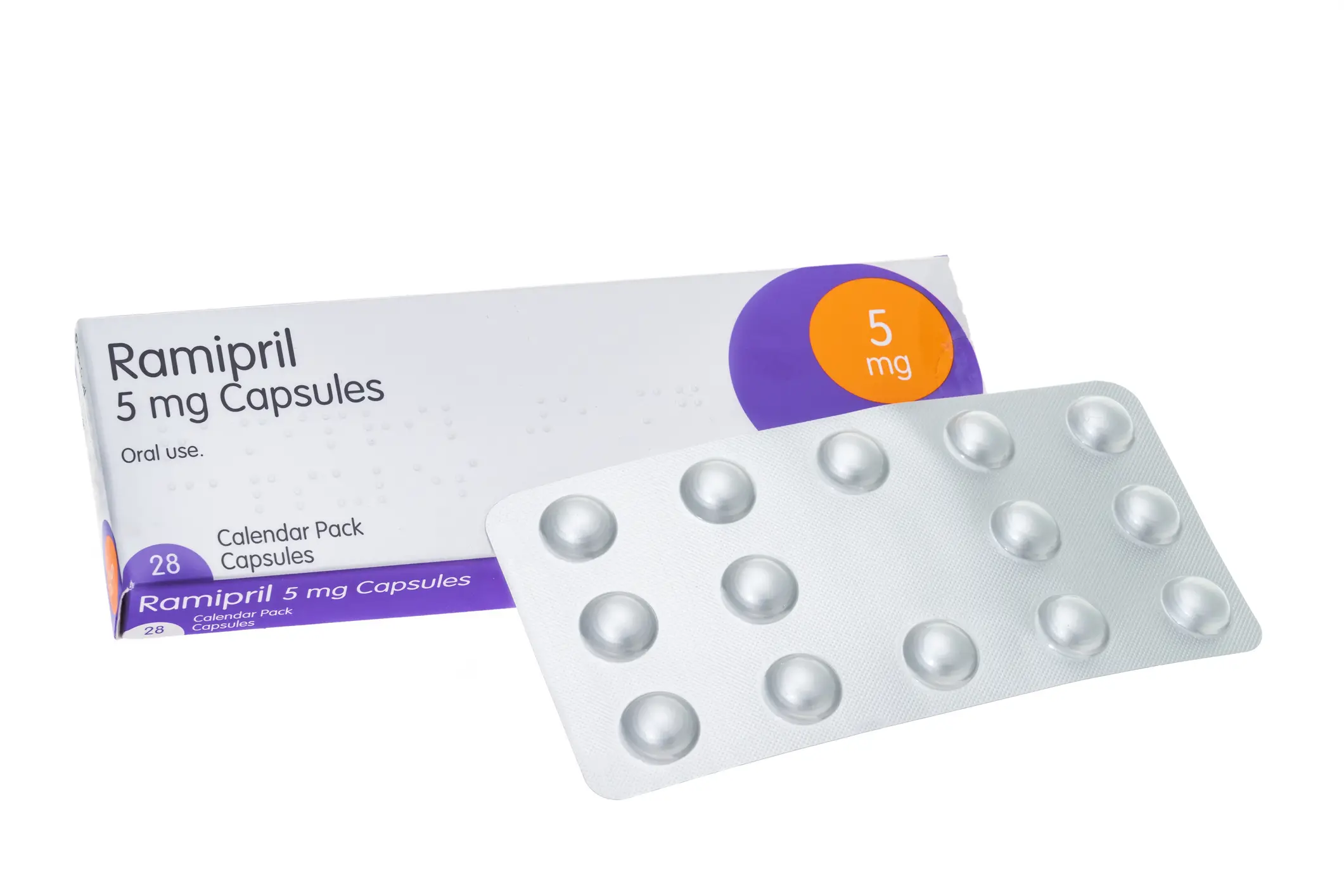
Produced by Crescent Pharma Limited, the affected medication is 5 mg capsules of Ramipril, with Batch Number GR164099 highlighted by the company as the potentially compromised group.
The issue appears to lie in the fact that this particular batch actually contains a completely different medication, with the blister strip actually containing Amlodipine 5 mg tablets instead.
This, thankfully, isn't as big of an issue as it might initially seem as both medications are designed to treat high blood pressure within patients, but there is still the risk of a concerning side effect among some individuals.
"The risk to patients of the accidental substitution of one widely used blood pressure medication for another is low," explains the Medicines and Healthcare products Regulatory Agency (MHRA) in a new statement, adding that "the most likely side effect would be dizziness related to low blood pressure."

As to what you should do if you've been prescribed Ramipril, MHRA Deputy Director Shareen Doak has outlined the steps you should follow:
"If you take Ramipril, check the packaging for batch number GR164099," she illustrated. "The batch number and expiry date information can be found on the outer carton.
"If you have received this batch, check that the medication name on the carton matches the blister strips inside. If the carton contains blister strips that are labelled as Amlodipine 5 mg tablets, contact your dispensing pharmacy. If the carton contains blister strips that are correctly labelled as Ramipril 5 mg Capsules, you do not need to take further action."
She adds that anyone experiencing side effects after discovering the mixed-up tablets should seek medical advice as soon as possible, taking the leaflet that came with the tablets along to your next doctor's appointment.
"Both medications are used to treat high blood pressure, however because your body may not be used to a different type of medicine, your blood pressure may become lower than normal, and you may experience dizziness because of taking amlodipine," Doak continued.